
Multiply each atom’s atomic mass by the number of atoms in the compound.Īs a result, it CaCO 3 has a molar mass of 100 g/mol. 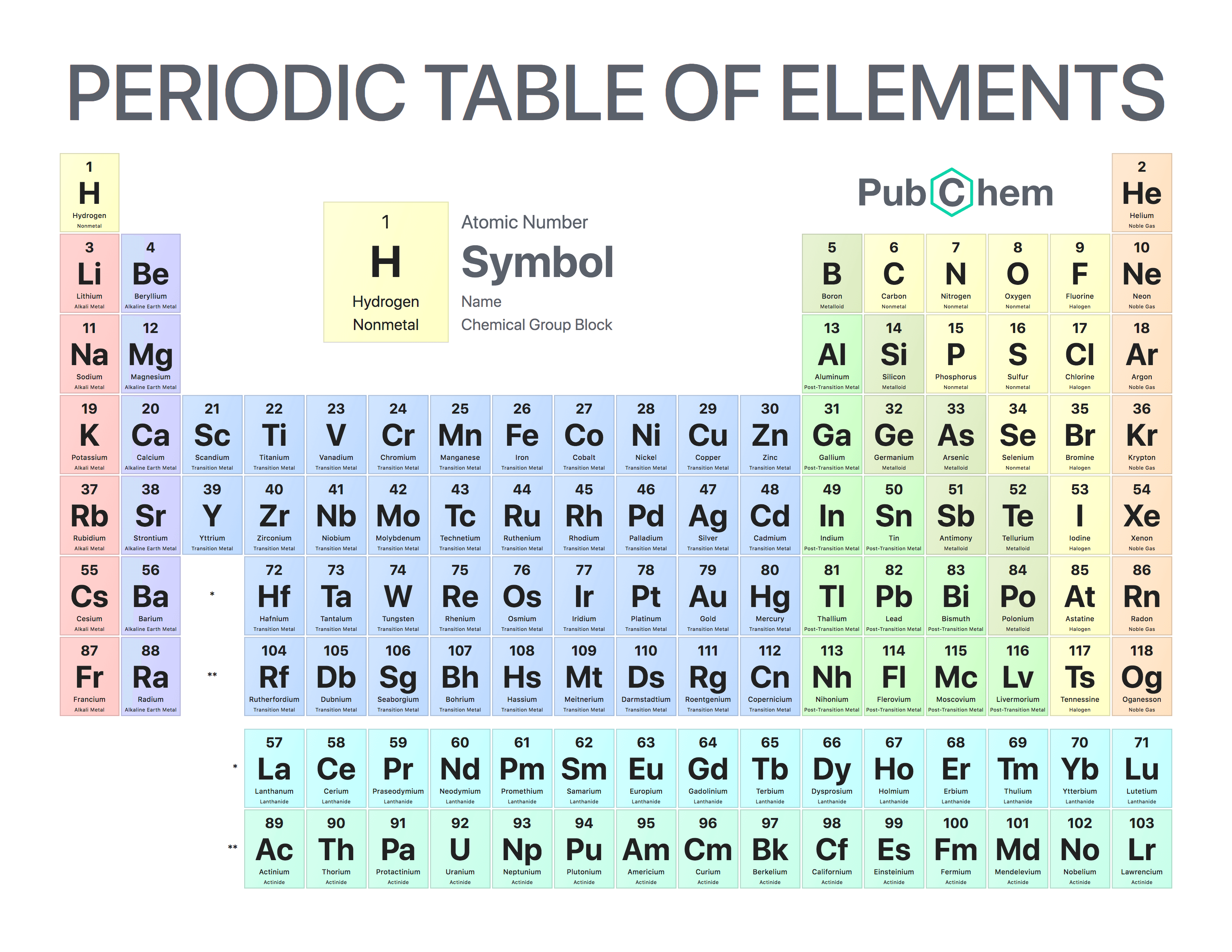
To get the molecular mass of CaCO 3, we must first determine the atomic masses of carbon (C), calcium (Ca) and oxygen (O). The total will be the compound’s molecular mass. For each atom in the molecule, add these values together.The subscript following the element symbol in the molecular formula represents this number. 
Multiply the atomic mass of each element in the molecule by the number of its atoms. The atomic weights listed on this Table of Elements have been rounded to the nearest whole number.Determine the atomic mass of each element in the molecule using the periodic table.Find out what the molecule’s molecular formula is.The development of the electronic theory of. and all the isotopes of an element occupy the same place in the periodic system in spite of their differences in atomic weight. To determine molecular mass, we’ll first go through the steps below. Mendeleyev’s periodic table of 1869 contained 17 columns, with two nearly complete periods. Identifying Elements the alkali metal with 11 protons and a mass number of 23 the noble gas element with and 75 neutrons in its nucleus and 54 electrons in. To calculate the molar mass of a specific substance, we will utilise the atomic masses of the constituent elements from the periodic table. To determine the molar mass, we must first comprehend the procedure for calculating the molar mass of any compound, after which we will proceed with the supplied substance using this procedure. The molecular weight of elements refers to periodic table of elements. Because various isotopes of an element are present in distinct molecules of the same substance, their molecular weights might vary. Molecular weight calculator can be used to calculate the average molecular weight (MW) of a molecule by entering a chemical formula. The molecular mass (m) of a molecule is measured in daltons (Da or u). As a result, the molecular mass of CaCO 3 is 100 u or 100 g/mol. There is just one carbon atom in the universe. We know that mass increases as the atomic number increases, and depends primarily on the number of protons and neutrons in the atoms. Calcium has an atomic mass of 40u, and Oxygen has an atomic mass of 16u and Carbon has an atomic mass of 12u.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
